483Dec 16, 2022
483 Document: Terumo Cardiovascular Systems Corporation
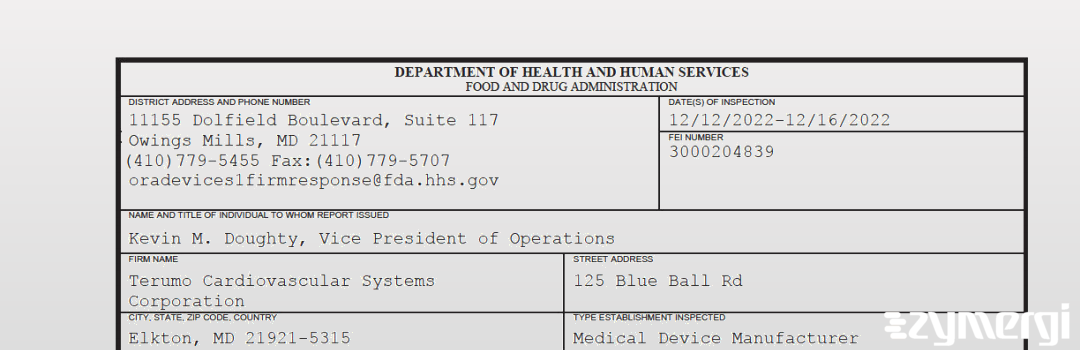
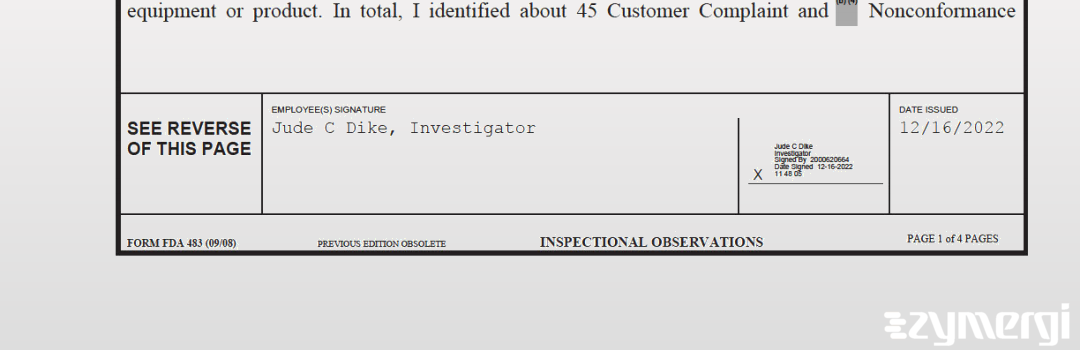
On Dec 16, 2022, the FDA inspected Terumo Cardiovascular Systems Corporation's Elkton site. Explore the inspectional observations.


On Dec 16, 2022, the FDA inspected Terumo Cardiovascular Systems Corporation's Elkton site. Explore the inspectional observations.